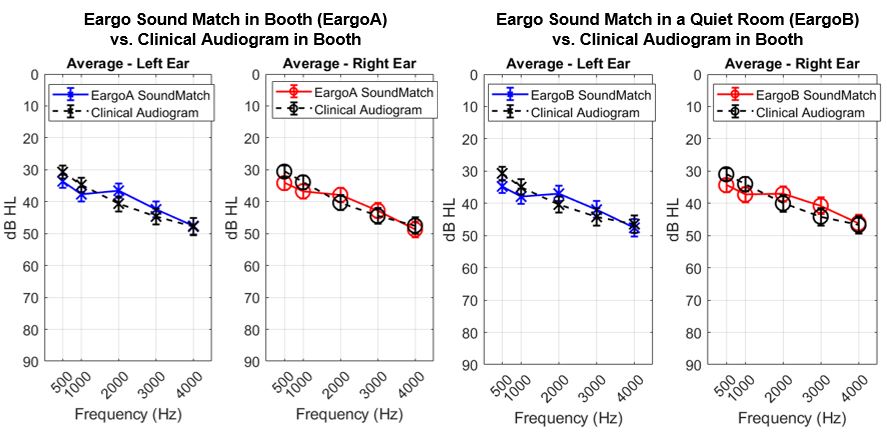
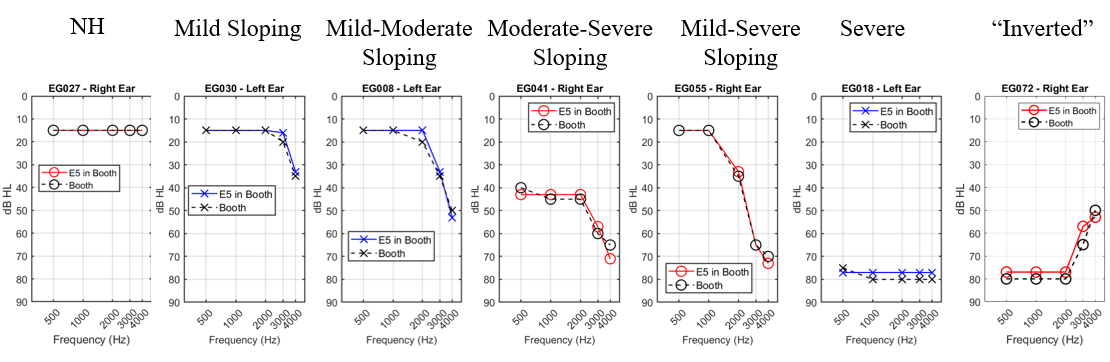
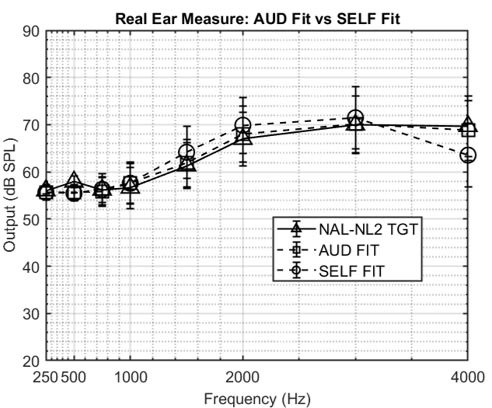
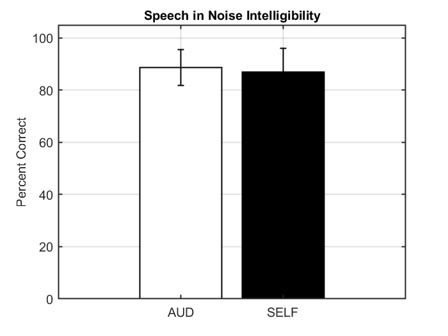
No, all OTC devices are not created equal. The FDA OTC Hearing Aid Final Rule created two distinct categories for hearing aids: prescription and OTC. There are broadly two kinds of OTC hearing aids: those with preset amplification levels with limited customization capabilities and those that are self-fitting.
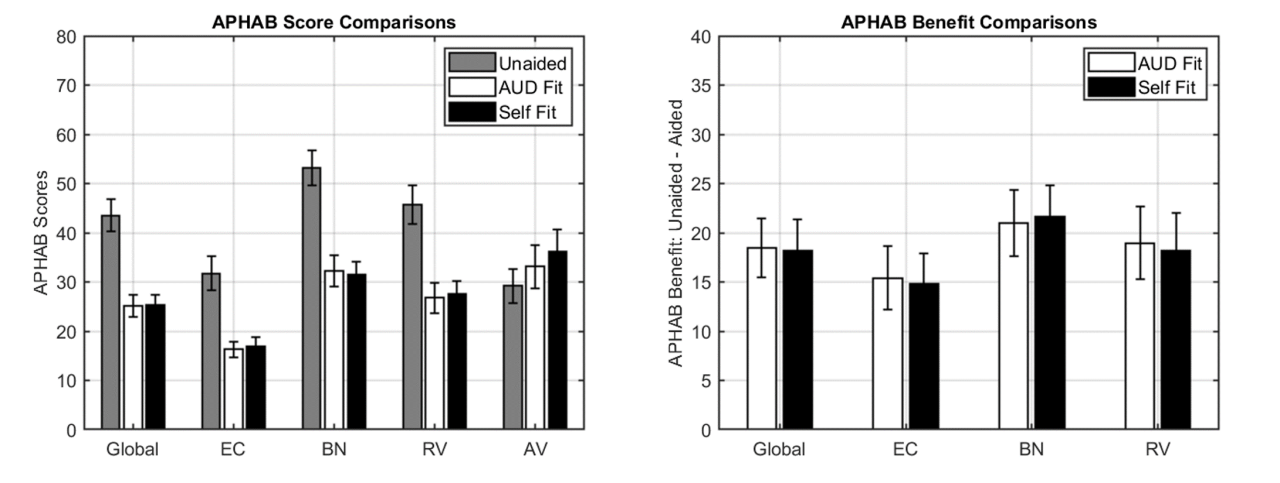
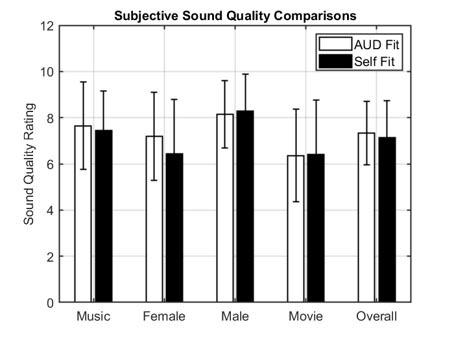
Self-fitting OTC hearing aids can be customized on an individual’s hearing loss. Self-fitting hearing aids require FDA 510(k) pre-market notification and clearance, which includes submission of clinical data to validate the effectiveness of the self-fitting strategy. Once reviewed and cleared by the FDA, these devices are considered FDA-cleared hearing aids.
Preset-based OTC hearing aids typically have preset settings that are designed to compensate for mild to moderate hearing losses. These devices have limited customization. These devices do NOT require the submission of clinical evidence to the FDA to demonstrate product efficacy. Once the manufacturer registers these devices with the FDA, the devices are considered FDA-registered hearing aids.
Eargo currently offers FDA-cleared self-fitting hearing aids (Eargo SE, Eargo 7, and Eargo 6) and an FDA-registered preset-based hearing aid (LINK by Eargo).